Methyl Benzoate Density G Ml
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name Methyl benzoate | |||
| Systematic IUPAC name Methyl benzenecarboxylate | |||
| Identifiers | |||
| CAS Number |
| ||
| 3D model (JSmol) |
| ||
| ChEMBL |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.002.055 | ||
| PubChem CID |
| ||
| UNII |
| ||
| CompTox Dashboard (EPA) |
| ||
| InChI
| |||
| SMILES
| |||
| Properties | |||
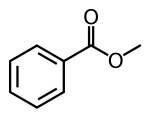
| Chemical formula | C 8 H 8 O 2 | ||
| Tooth mass | 136.150 thousand·mol−one | ||
| Density | 1.0837 thou/cmiii | ||
| Melting point | −12.5 °C (9.v °F; 260.vi K) | ||
| Humid bespeak | 199.6 °C (391.iii °F; 472.viii K) | ||
| Magnetic susceptibility (χ) | −81.95×10−six cmthree/mol | ||
| Refractive index (n D) | one.5164 | ||
| Hazards | |||
| NFPA 704 (burn down diamond) | 0 2 0 | ||
| Wink signal | 82 °C (180 °F; 355 K) | ||
| Safety data canvass (SDS) | ScienceLab MSDS | ||
| Related compounds | |||
| Related compounds |
| ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |||
Methyl benzoate is an organic compound. It is an ester with the chemic formula CviH5COtwoCH3. It is a colorless liquid that is poorly soluble in h2o, but miscible with organic solvents. Methyl benzoate has a pleasant smell, strongly reminiscent of the fruit of the feijoa tree, and it is used in perfumery. It also finds use every bit a solvent and every bit a pesticide used to concenter insects such as orchid bees.
Synthesis and reactions [edit]
Methyl benzoate is formed by the condensation of methanol and benzoic acid, in presence of a strong acid.[one] [2]
Methyl benzoate reacts at both the band and the ester, depending on the substrate. Electrophiles set on the ring, illustrated by acid-catalysed nitration with nitric acrid to give methyl 3-nitrobenzoate. Nucleophiles assault the carbonyl center, illustrated past hydrolysis with addition of aqueous NaOH to give methanol and sodium benzoate.
Occurrence [edit]
Methyl benzoate tin can be isolated from the freshwater fern Salvinia molesta.[iii] It is one of many compounds that is attractive to males of diverse species of orchid bees, which obviously assemble the chemic to synthesize pheromones; it is commonly used every bit bait to attract and collect these bees for study.[4]
Cocaine hydrochloride hydrolyzes in moist air to give methyl benzoate;[5] drug-sniffing dogs are thus trained to discover the smell of methyl benzoate.[half-dozen]
Uses [edit]
Non electric Estrus cost allocators. See: DIN EN 835.
References [edit]
- ^ Maki, Takao; Takeda, Kazuo. "Benzoic Acid and Derivatives". Ullmann'due south Encyclopedia of Industrial Chemical science. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_555. .
- ^ John McMurry (2008). Organic Chemistry, seventh Edition. Thompson - Brooks/Cole. ISBN978-1-4390-4972-three. . Page 623
- ^ Choudhary, MI; Naheed, Due north; Abbaskhan, A; Musharraf, SG; Siddiqui, H; Atta-Ur-Rahman (2008). "Phenolic and other constituents of fresh h2o fern Salvinia molesta". Phytochemistry. 69 (4): 1018–23. doi:10.1016/j.phytochem.2007.10.028. PMID 18177906.
- ^ Schiestl, F.P.; Roubik, D.Westward. (2003). "Odor Compound Detection in Male Euglossine Bees". Periodical of Chemical Ecology. 29 (1): 253–257. doi:x.1023/A:1021932131526. hdl:20.500.11850/57276. PMID 12647866.
- ^ Dejarme, Lindy E.; Gooding, Rachel East.; Lawhon, Sara J.; Ray, Prasenjit; Kuhlman, Michael R. (1997). "Formation of methyl benzoate from cocaine hydrochloride nether different temperatures and humidities". In Works, George; Rudin, Leonid I; Hicks, John; et al. (eds.). Proceedings of SPIE. SPIE Proceedings. Vol. 2937. p. nineteen. doi:10.1117/12.266783.
- ^ Waggoner, L. Paul; Johnston, James M.; Williams, Marc; Jackson, Jan; Jones, Meredith H.; Boussom, Teresa; Petrousky, James A. (1997). "Canine olfactory sensitivity to cocaine hydrochloride and methyl benzoate". In Works, George; Rudin, Leonid I; Hicks, John; et al. (eds.). Proceedings of SPIE. SPIE Proceedings. Vol. 2937. p. 216. doi:10.1117/12.266775.
Methyl Benzoate Density G Ml,
Source: https://en.wikipedia.org/wiki/Methyl_benzoate
Posted by: howardablightmed.blogspot.com




0 Response to "Methyl Benzoate Density G Ml"
Post a Comment